Fission yeast cells are rod shaped and divide by medial fission. The division cycle is quite rapid, with a generation time of S. pombe between 2 and 4 hours . It’s easy and inexpensive to grow fission yeast and manipulate the cells in the laboratory.
S. pombe is a popular system for studies of cell growth and division, partly because of its regular size. Additionally, its chromosome structure is a good model for human chromosomes. Fission yeast shares numerous features with human chromosomes including large and complex centromeres and replication origins, “typical” heterochromatin,and small ncRNA (miRNA) regulation; these features are missing, or different, in budding yeast.
A cartoon of the fission yeast life cycle is shown below. Vegetative, or mitotic growth, is diagrammed on the right. In exponential phase, G2 phase is particularly long, with G1, S phase (DNA replication) and M phase (Mitosis) each taking about 10% of the division time. Generally fission yeast is a haploid, with diploid growth only a transient stage in sexual differentiation. However, in the laboratory, diploids can be recovered and grown in the vegetative cycle.
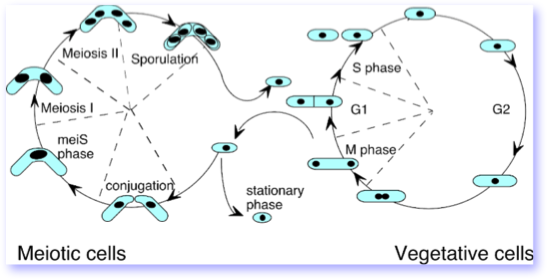 On the left, the cartoon diagrams the specialized meiotic cycle that results after two cells conjugate, forming a transient diploid, or zygote, that proceeds through meiosis to produce four spores . These are essentially the lifeboats of a unicellular organism, packaged neatly into a tetrad ascus. The ability to undergo sexual differentiation and exchange, or recombine, genetic information, makes fission yeast a very powerful model genetic system.
On the left, the cartoon diagrams the specialized meiotic cycle that results after two cells conjugate, forming a transient diploid, or zygote, that proceeds through meiosis to produce four spores . These are essentially the lifeboats of a unicellular organism, packaged neatly into a tetrad ascus. The ability to undergo sexual differentiation and exchange, or recombine, genetic information, makes fission yeast a very powerful model genetic system.
Aside: FACS analysis
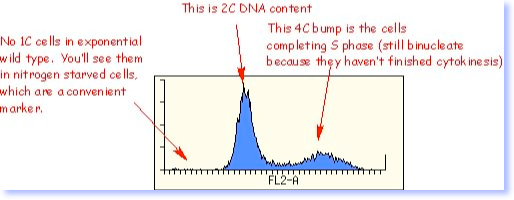
People doing flow cytometry on exponentially growing S. pombe cells are puzzled that there is only one peak, corresponding to 2C DNA content. Fission yeast DOES have a G1 phase, but it is short (about 10% of the cell cycle, as noted above). And, the timing of cytokinesis and the timing of the nuclear division cycle are somewhat out of synch. This means that after nuclear division occurs, the nuclei go through G1 and enter S phase before the cells complete cytokinesis. As far as a flow cytometer is concerned, a binucleate cell with two 1C (G1) nuclei looks the same as a uninucleate cell with a single 2C (G2) nucleus. There are a couple of ways around this that can give you a true G1 peak. You can perform FACS on prepared nuclei (ghosts). Or, you can use 2-D analysis by comparing pulse area to width. This discriminates single nuclei from double nuclei. If you eliminate doublets by appropriate gating, you gain significant additional information. Both these methods were developed by Erik Boye’s lab in Oslo.
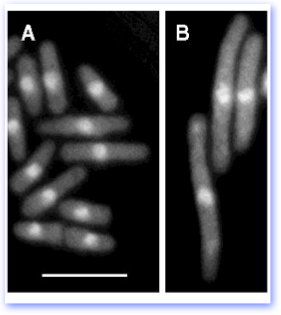
Isolating mutants Since fission yeast are typically haploid, it is easy to isolate recessive mutations. Of course, if you mutate an essential gene, the cells would be dead. Thus, as in other microbial systems, pombe geneticists make use of conditional alleles. The most frequent condition is high temperature, in which a protein is inactivated at 36°, although cold temperature or conditional gene expression can also be used.
The cells shown in panel A of the figure are wild type cells, viewed under the microscope and treated with a fluorescent dye (DAPI) that stains the DNA in the nucleus. The nuclei are the bright blobs in the center of the cells. At the bottom of the panel, you can see a cell that has divided, with two nuclei and a medial septum. The bar indicates scale and is 10 microns long.
If the cells have a mutation that prevents the normal progression of the cell cycle, they become very elongated. These mutants are called cdc mutants, for cell division cycle. This occurs because the cells can continue to grow but are unable to divide (panel B).
In this particular example, taken from an early paper from our group, cells contain a temperature sensitive mutation in the cdc19 gene, encoding the Mcm2 subunit of the MiniChromosome Maintenance complex required for DNA replication. THey have been shifted to the restrictive temperature of 36°C. The mutation blocks cell cycle progression during S phase and prevents the cells from going on to divide, but they can still undergo macromolecular synthesis and cell growth, hence the elongation.
Go to the next pages for information on DNA replication , chromosomes, and meiosis.