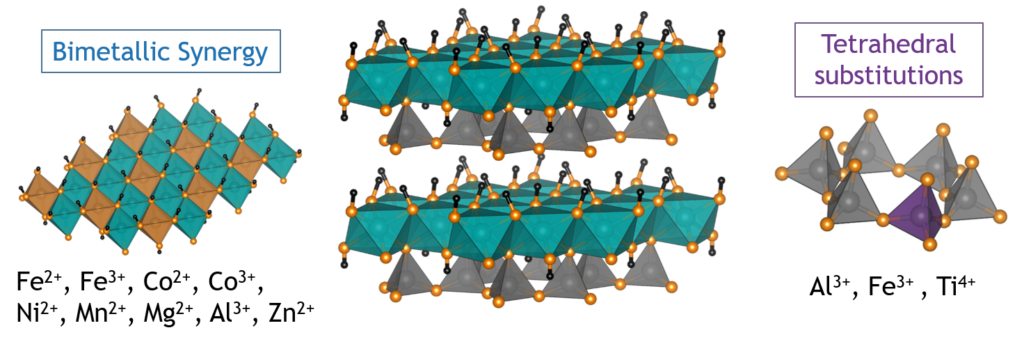
Heterogeneous catalysts provide simple, separation-free ways to manipulate the selectivity of organic reactions and are used in nearly every industrially relevant chemical transformation. Homogeneous oxidation catalysis relies on harsh chemicals like chromic and nitric acids that are both extremely toxic and heavily regulated environmental pollutants. Our group is working to develop new heterogeneous catalysts that can utilize molecular oxidation as a way to developer greener approaches for the chemical industry that reduce the use of these toxic chemicals. Much of our work to-date has focused the phyllosilicate family of minerals, illustrated above,where there are sheets of silicate groups bonded to sheets of transition metals. We have found the catalytic properties of these materials can by activated by heating at elevated temperatures to remove some of the hydroxyl groups from the surface of the sheets to expose coordinatively unsaturated metal sites where molecules can react. We are using local structure probes like Pair Distribution Function (PDF) analysis and Extended X-ray Absorption Fine Structure (EXAFS) spectroscopy to study how changes to the surface of the material affect the catalytic properties.
Contact
Brent C. Melot
Professor of Chemistry, Chemical Engineering, and Materials
Department of Chemistry
SGM 213 3620
McClintock Avenue Los Angeles, CA 90089-1062