CNT3D combines several key platform technologies for drug discovery and development in its core labs. These synergistic technologies are then made available to researchers through collaborations across both USC campuses and the broader SoCal community.
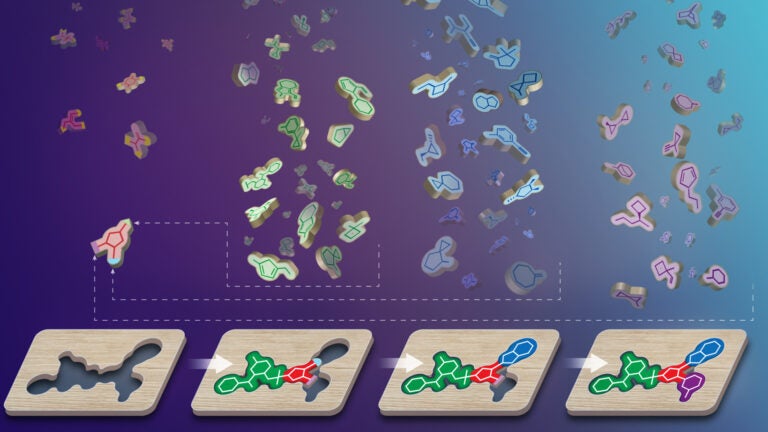
V-SYNTHES
Developed in Dr. Katritch’s lab (Nature 2021), V-SYNTHES is a giga-scale structure-based virtual screening technology for hit and lead discovery. This approach, currently expanded to screening more than 3 trillion on-demand compounds, has been successfully applied in hit and lead identification for more than a dozen clinically relevant targets in various collaborations. In collaboration with Drs Aiichiro Nakano and Yan Liu, we are working on a new V-SYNTHES versions with additional AI capabilities.
Structural Pharmacology
State-of-the-art structure determination XFEL and Cryo-EM technologies, developed in the labs of Dr. Cherezov and Dr. Gati, make it possible to determine high-resolution structures of key drug targets with a variety of ligands and drugs and in different conformational states in a matter of weeks, or even days. This provides a solid platform for computational screening and design, as well as rational optimization of the identified hits and leads. The most recent structures from Cherezov and Gati labs include such important drug targets as GPCRs and membrane transporters (Lin et al 2023 Cell discovery. 9(1):23; Motiwala et al 2022 Nature 606(7915):820-6.; Shaye et al 2020 Nature 584:298-303; Michaelian et al (2021 Nature Commun. 12(1):2971.)
Prodrug Design and Medicinal Chemistry
Novel prodrug technology developed in the Dr. McKenna lab (Lloyd et al. 2022. Antiviral Res. 199:105275) targets selective inhibition of DNA and RNA replication, yielding a highly effective oral compound against viral infections (in this instance, shingles). The technology is widely adaptable to create more effective drugs against other viruses, including coronavirus (2023 Int. Conf. Antiviral Research, Lyon, March 2023: Abs 562, 604, 610, 611). McKenna lab has also created and validated a first-of-kind epigenetic antifungal drug concept targeting a new class of fungal protein targets called bromodomains, implemented by a screening campaign using V-SYNTHES with the Katritch lab (Wei et al, 2023 bioRxiv. 2023.02.03.527073. doi: 10.1101/2023.02.03.527073). Other delivery technologies for bone-targeting drugs are also under development (Okawa et al 2022, Elife doi: 10.7554/eLife.76207; Okawa et al 2022, Nature Commun Med 2:112. Epub 2022/09/10. Doi: 10.1038/s43856-022-00172-x.
Click Chemistry and Analytical Chemistry
Developed in Dr. Fokin lab, the click chemistry approaches have been a major driver in drug discovery. A new click-based combinatorial libraries have been validated for cannabinoid receptors in collaboration with the Katritch lab, yielding highly potent hits from the screening of 140 Million compounds (Grotsch et al 2023, under review.). Another highly relevant technology for drug discovery is the design and screening of specific covalent proximity probes based on click chemistry, either as drug candidates (Fokin and Katritch patent application US 2021/0061814) or as the initial screening of challenging targets.
Systems Biology
Systems biology is a key modern tool in both translational medicine and target discovery and validation: It involves the integration of quantitative experimental studies, mathematical modeling, and computational analyses to tackle complex biological questions. Dr. Stacey Finley has long-standing expertise in building mechanistic, experimentally-validated systems biology models to study cell signaling, metabolism, and drug therapeutic effects in cancer and other diseases.
Quantitative Structural Cell Biology
Dr. Kate White lab is focused on developing and applying methods for 3D single-cell imaging to better understand cellular rearrangements, also helping to establish infrastructure for archiving and sharing whole cell structural information to enable computational modeling of complex cellular functions such as glucose-stimulated insulin secretion. The goals are to extend structure-based drug discovery by linking cell structure and function in more depth.