Emerging Investigator
USC Dornsife’s Matthew Pratt, assistant professor of chemistry and biological sciences, has been named a 2013 Emerging Investigator by ChemComm, a leading chemical sciences journal published by the Royal Society of Chemistry.
The distinction recognizes top chemical sciences researchers in the early stages of their careers. He was among 94 researchers from throughout the world selected for the honor in the journal’s recent Emerging Investigators issue.
The ChemComm edition also published a paper authored by Pratt focusing on the development of synthesis and characterization of analogs of sugars that can be fed to cancer cells. This process allows the direct visualization of the modification of proteins.
Pratt’s research studies posttranslational modifications of proteins and how these modifications promote cancer or can affect neurodegenerative disease.
“We are trying to identify new protein targets that we can then develop drug-like molecule inhibitors against,” Pratt said. “We know that cancer cells alter their own metabolism quite radically from normal adult cells. So we thought since much of the alteration is in glucose metabolism, that may factor into this modification.”
Experiments performed by Pratt and his team appear to confirm that this is the case.
“We used these tools to look at the individual steps between cancer metabolism and the modification. In the process, we found an enzyme that is upregulated in cancer but not in normal cells,” he said, referring to an enzyme that regulates some aspect of a cell or body up upping the production of something.
“So now the next step is to attempt to target that with some inhibitors in order to see if we bring the activity levels down whether the cancer cells will no longer form tumors or survive.”
If that proves to be the case, Pratt and his team will then try to validate the fact that these enzymes may be important drug targets in battling cancer.
Pratt has published eight papers in leading scientific journals since joining USC Dornsife four years ago.
“It’s nice to be recognized for your early work,” Pratt said. “For me, the most fulfilling part of the job is teaching the craft of doing science and learning new things that no one knew before.”
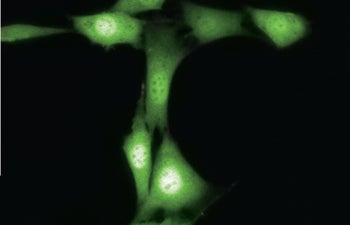
The Matt Pratt laboratory researches O-GlcNAc modification in cancer cells, including HeLa cells, shown here expressing the green fluorescent protein. Image courtesy of Matt Pratt and Hubert Lau.
Pratt’s research has earned him a prestigious grant from The Michael J. Fox Foundation (MJFF) to continue his investigation of potential treatment therapies for Parkinson’s disease, a degenerative neurological disorder.
“Right now, there are no therapies available that slow the progress of Parkinson’s disease in the brain — the existing therapies simply deal with symptoms,” he said.
Parkinson’s disease affects one in 100 people over age 60, though its cause is unknown. Recent research indicates that at least one million people in the United States have Parkinson’s disease. Symptoms include shaking, rigidity, slowness of movement and cognitive impairment ranging from mild memory difficulties to dementia.
Co-founded by Michael J. Fox, the Golden Globe-winning actor diagnosed with Parkinson’s in 1991, and Deborah Brooks, the foundation is dedicated to finding a cure for Parkinson’s disease through an aggressively funded research agenda. MJFF’s diverse research portfolio includes significant investments to better understand a specific protein called alpha-synuclein, which plays an important role in the advancement of the disease.
Alpha-synuclein proteins form toxic aggregates called Lewy bodies in the brains of Parkinson’s patients. These deposits, the cardinal neuropathological feature of the disease, were discovered in the early 20th century. However, only recently has it been shown definitively that the aggregates kill neurons, and when injected into laboratory mice, the aggregates spread in the same progressive fashion as in humans. The alpha-synuclein deposits accumulate in the center of brain cells until they eventually spread throughout the brain, killing more cells.
“Our research strategy is based on the fact that these proteins can be modified endogenously by posttranslational modifications,” said Pratt. Posttranslational modifications are a step in protein biosynthesis, and the particular amino acids and enzymes involved in this process determine the final protein’s function.
“The alpha-synuclein is sort of a scaffold for a wide variety of modifications, but for this grant we are focusing on one in particular.”
This modification is a type of carbohydrate called O-GlcNAc that is attached to the alpha-synuclein protein through the side chains of its amino acids, specifically to the amino acid threonine 72. The modification is strongly linked to glucose metabolism, and the biosynthesis of glucose into other things is usually impaired in Parkinson’s patients, yielding presumably less of this type of modification.
Pratt expects that increasing this modification may block the aggregation of alpha-synuclein in brain cells, and this grant is a first step aimed at directly testing that question.
“There are medical research companies interested in using small molecules, or inhibitors, to increase levels of this posttranslational modification in Alzheimer’s patients as a way to prevent protein aggregation, and we’re interested in seeing if that could also be a strategy for Parkinson’s.”
As protein chemists, Pratt and his research team are taking a less clinical and more foundational, biochemical approach. Using biophysical techniques, they will make the modification on the protein in a test tube to evaluate its effect on protein aggregation in the lab.
If it successfully blocks aggregation, this research may lead to partnerships with companies to develop inhibitors that can serve as a new treatment for Parkinson’s, one that is not merely palliative but actively inhibits the progress of the disease.
Pratt’s research is supported by the Damon Runyon Cancer Research Foundation and the California-based Concern Foundation.